FERTILITY ROAD ARTICLE
Looking for Answers After a Failed Transfer
Dr. Elizabeth Barbieri of Oregon Reproductive Medicine helps explain the leading causes of failed transfers and miscarriages in IVF treatments.
A failed transfer is understandably emotionally difficult time for patients. Multiple failed transfers compound this stress and can be extremely costly as well. When a failed transfer occurs, patients are left questioning “Why didn’t it work?” “Is something wrong with me?” and looking for answers.
 Where to Look After a Failed Transfer?
Where to Look After a Failed Transfer?
To try and develop a treatment strategy that has the best chance of success on a subsequent attempt, we need to look for answers in the right places. I often hear questions about the role of Natural Killer cells and the role of a good uterine lining in the context of failed transfers, so it is important to address these concerns. However, the most common reason an embryo fails to implant is that the embryo is chromosomally abnormal i.e. is missing a chromosome or has an extra chromosome and cannot develop into a viable pregnancy and a healthy baby.
Natural Killer Cells are white blood cells that play an essential role in the body’s immune system by targeting and destroying foreign cells. They are present in the blood and also in the uterine lining. It has been suggested that Natural Killer cells play a part in pregnancy and that elevated levels may cause a miscarriage by rejecting an embryo. While some physicians and clinicians subscribe to this theory and point to some studies as evidence, a detrimental role for elevated Natural Killer cell levels has yet to be conclusively demonstrated and there is evidence that Natural Killer cells in the uterus operate differently to those in the rest of the body. Complicating matters is that there is no standardized way to measure Natural Killer cell levels or agreement on what comprises elevated levels.
Treatments that seek to suppress Natural Killer levels are called immunosuppressive therapy and can include steroids, intravenous immunoglobulin (IVIg), tumor necrosis factor-a (TNF) blocking agents and intralipid infusions. These treatments may have important side-effects and in the UK the sector regulator, the Human Fertilisation & Embryology Authority, has not licensed these therapies for fertility treatment and does not support Natural Killer cell theory. In the USA there is similarly a lack of agreement on the effectiveness of treatments that may be available.
Development of the Uterine Lining prior to a transfer is a critical factor for implantation, which is a pre-requisite for the further development of a pregnancy. We assess the uterine lining (endometrium) by measuring its thickness (endometrial thickness or EMT) with a transvaginal ultrasound. It is generally accepted that a “thick” lining of 7mm or more – one that is akin in some ways to plush velvet – will be the best for implantation. Accurate measurement of the of the uterine lining thickness to ensure its optimum development prior to transfer requires high-resolution ultrasounds and skilled manipulation of the transducer, as the measurement of the same lining by individuals of differing skill or using different resolution equipment may have significant variation. Studies indicate that there is a significantly lower chance of implantation and a clinical pregnancy when the uterine lining has a thickness of less than 7mm, so small variations in measured thickness can be important. Of course, not all clinical pregnancies will continue to develop further and it is necessary to consider other relevant factors if a transfer fails.
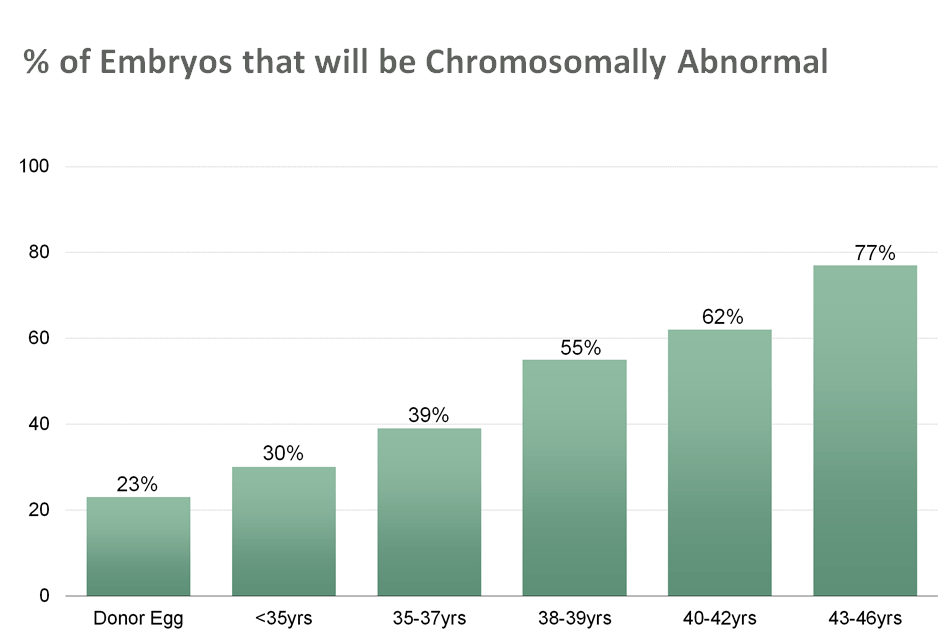
Chromosomally Abnormal Embryos is well documented as the leading cause of failed implantation and miscarriage in both IVF and naturally conceived pregnancies. Studies of first trimester miscarriages tell us that 60% or more are the results of chromosomally abnormal embryos, and studies of still-births indicate chromosomal abnormality in some 5-10% of cases. Many IVF patients will have heard about the grading of embryos based on the number of cells at the blastocyst stage, how healthy they look, and their rate of development. This morphological assessment is important; however, whether an embryo is chromosomally normal (euploid) and possesses the correct number of complete chromosomes (46), or does not and is chromosomally abnormal (aneuploid) cannot be determined by a visual inspection and this critical factor is not part of the grading system for embryos that many patients hear about.
A recent study by Calpabo et al in Human Reproduction analyzing 956 blastocysts demonstrated that 44% of “excellent” grade blastocysts and 61% of “good” grade blastocysts were actually chromosomally abnormal.
What are and What Causes Chromosomally Abnormal Embryos?
Chromosomal abnormalities in embryos can occur in two basic forms. There can be numerical abnormalities (an incorrect number of chromosomes) and structural abnormalities (translocations which are rearrangements of parts of different chromosomes; or duplications or deletions of a part of a chromosome). A normal embryo should have 46 chromosomes, 23 pairs. The first 22 pairs of chromosomes are called the autosomal chromosomes. The last 2 are the sex chromosomes and should either be female (XX) or male (XY). A missing chromosome is called a monosomy; an extra chromosome is called a trisomy. Almost all cases of a missing or extra autosomal chromosome and most structural abnormalities would cause an embryo to not implant or to later miscarry. Some chromosomal abnormalities such as Turner Syndrome (monosomy X) or Downs Syndrome (trisomy 21) may develop into pregnancies but may result in a baby being born with these conditions.
Chromosomal abnormalities principally arise during the process of cell division. There can be errors during the division of the reproductive cells (meiosis) that leads to the mature egg or sperm, or errors during the division of the embryonic cells (mitosis) while the embryo is developing. Problems during meiosis result in eggs or sperm with chromosomal abnormalities that are passed on to the embryo. Errors in mitosis after fertilization can be replicated as the embryo grows to the blastocyst stage when it may be selected for transfer. Chromosomal abnormalities may arise in the embryo post transfer through errors in mitosis, and in these cases, external factors may play a part.
Chromosomally abnormal eggs are the leading cause of chromosomal abnormalities in embryos, and age – the proverbial “biological clock” – is the leading indicator of the chance that embryos formed from a woman’s eggs will be chromosomally normal or abnormal. Because of the fundamental difference in the way egg and sperm are made, age does not appear to play a significant part in sperm being chromosomally abnormal, although some recent studies indicate that male age does seem to play a part in higher incidence of other disorders.
 What is the Impact of Maternal Age and Other Factors?
What is the Impact of Maternal Age and Other Factors?
While we know conclusively that the rate of chromosomal abnormality in her eggs increases as a woman ages, we don’t yet fully understand the underlying cause(s). It is likely that several factors may each lead to chromosomally abnormal eggs and therefore abnormal embryos. In basic terms though, a woman is born with all of the eggs – albeit in an immature state – she will ever have, and unlike sperm, a woman’s body does not create new egg cells during her lifetime. So a woman of 25, 35 or 45 years old has eggs of the same age. As they age, the ability of a woman’s immature egg cells to grow into mature eggs without developing chromosomal abnormalities decreases. The rate of this decline begins to accelerate significantly from the age of 35.
At a basic biological level, with age, the chromosomes in a woman’s eggs lose their ability to properly segregate and replicate during the process of maturation. Studies indicate that reduced levels of a protein which is essential for biding the chromosomes together and segregating correctly during the maturation process may be the main factor. This chromosomal “cohesion” is established during fetal development and needs to remain functional as the egg ages. Other studies have found that the chromosomes in older eggs are significantly more likely to not “line-up” correctly, increasing the risk of uneven chromosome replication as an egg matures.
The cumulative impact of oxidative stress on the eggs (the same basic factor that leads to other age-related problems elsewhere in the body), as well as possible age-related hormonal changes that affect the process of an egg cell’s maturation may be root causes. In addition, exposure to certain environmental toxins such as bisphenol A (BPA) and cigarette smoking can lead to chromosomally abnormal eggs. It is plausible that other lifestyle factors may play a role for some women, but age explains the overwhelming incidence of chromosomal abnormalities and impacts all women.
Aside from age-related and other external factors, some women may also be genetically predisposed to have a higher rate of chromosomally abnormal eggs than normal for their age. For example, women with fragile X premutation are at risk of premature menopause, have fewer eggs and more chromosomally abnormal eggs than other women their same age. Additionally, one recent study proposed that a problem with the gene which regulates egg development may contribute to some women experiencing a higher than expected rate of abnormal embryo formation.
What are the Treatment Options?
The key to minimizing the risk of a failed transfer resulting from a chromosomally abnormal embryo is to only transfer an embryo with the correct number of complete chromosomes. The challenge it that chromosomally abnormal embryos often appear perfectly normal, even to the most experienced embryologist.
Preimplantation genetic screening (PGS) was developed in the early 1990s to help identify chromosomally abnormal embryos. Early PGS technologies only screened a limited number of chromosomes. The most advanced and precise form of PGS technology in use since about 2015 is called Comprehensive Chromosome Screening (CCS) with Next Generation Sequencing (NGS). CCS screens all 23 pairs of chromosomes so provides a complete picture. The most common prior-generation PGS technology still in use in many IVF labs and which many IVF patients may hear of is Comparative Genomic Hybridisation (referred to as CGH, aCGH or array CGH). Both CCS with NGS and CGH test all 23 pairs of chromosomes, but given its higher resolution CCS with NGS appears better at detecting both structural as well as numerical abnormalities.
The impact of this type of testing on the embryos prior to transfer can be dramatic and allows women up to the age of about 43 undergoing IVF to benefit from the same level of success rates as much younger women who would normally have significantly higher chances of success due to lower rates of chromosomally abnormal embryos.
Above a certain age, however, the chance of having chromosomally normal embryos which PGS testing can isolate becomes negligible, and which continued attempts at IVF using their own eggs will not overcome. Therefore for women in their 40’s the consideration of donor eggs as a viable option becomes an important one. Women in this age range can successfully carry a pregnancy a donor-conceived pregnancy without complications. Egg donors that are well-screened and in their 20s provide a high chance of having chromosomally normal embryos and therefore significantly increase the chances for a successful transfer and a healthy baby.